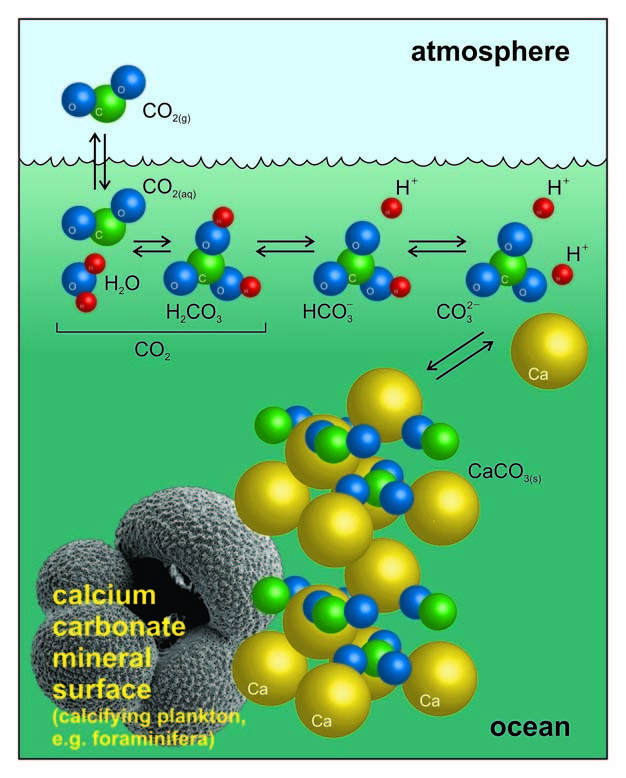
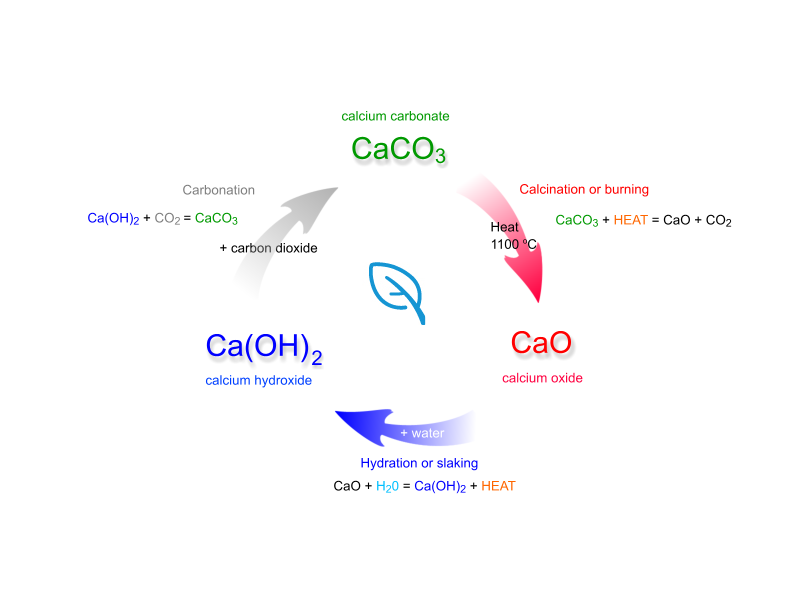
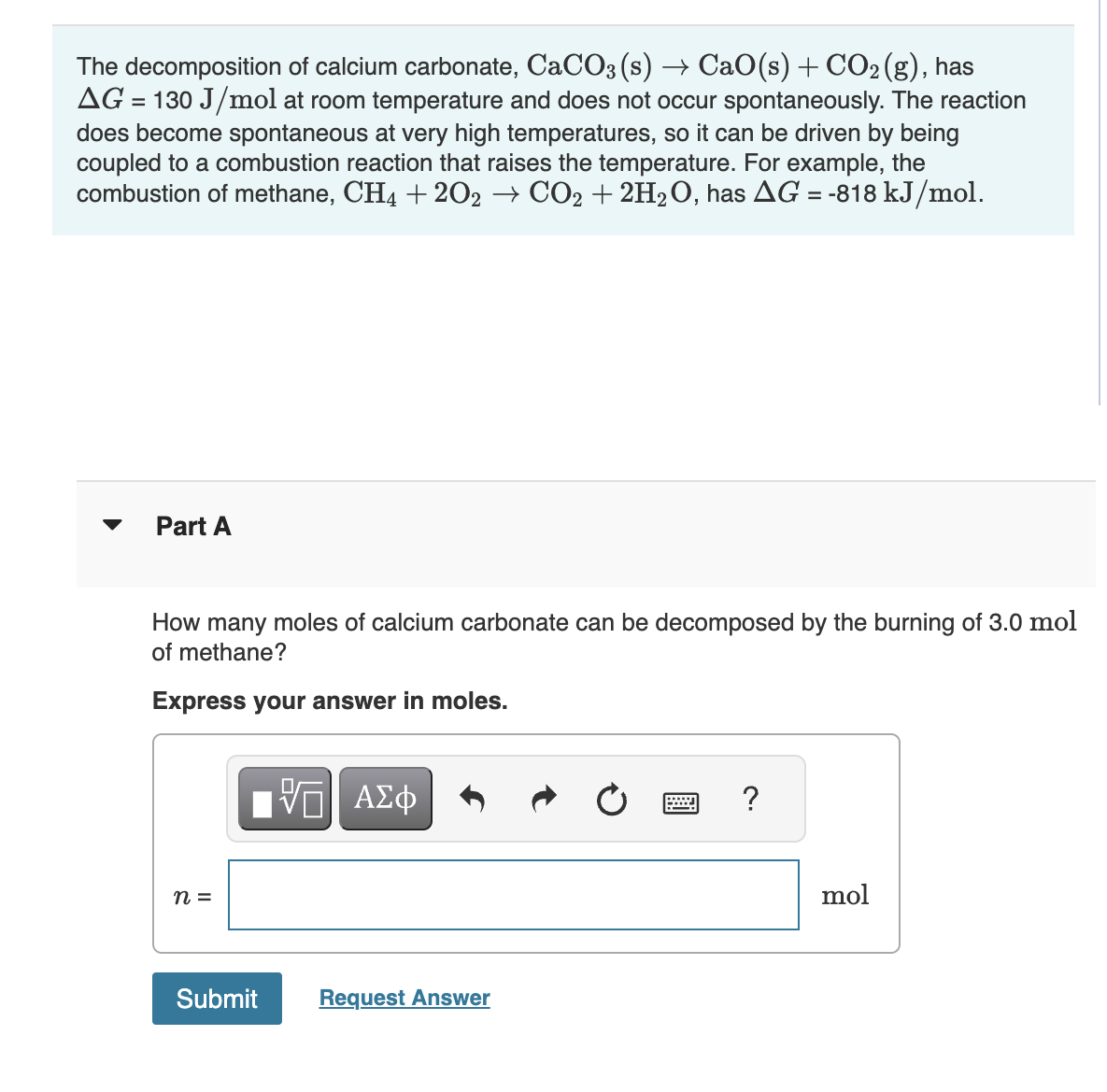
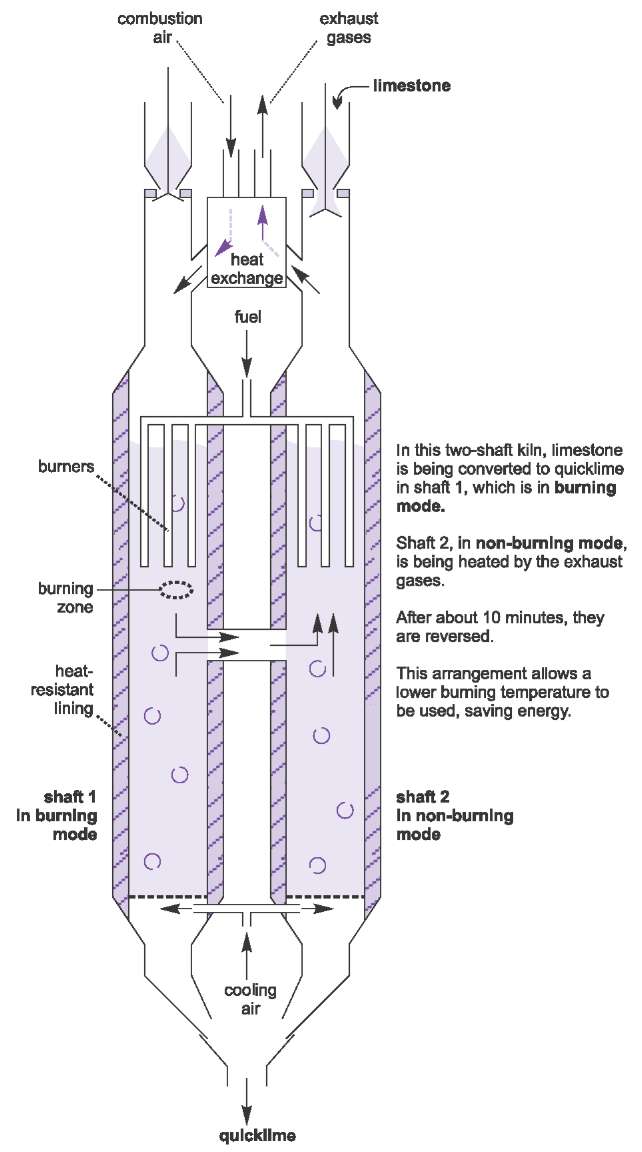
![Principles of agricultural chemistry [microform] . sea water is decomposed,setting carbon dioxide free, and the calcium carbonate is used bythe animal to form its shell. Ca(UCO,)., = CaCOg + HoO -f Principles of agricultural chemistry [microform] . sea water is decomposed,setting carbon dioxide free, and the calcium carbonate is used bythe animal to form its shell. Ca(UCO,)., = CaCOg + HoO -f](https://c8.alamy.com/comp/2CJ846R/principles-of-agricultural-chemistry-microform-sea-water-is-decomposedsetting-carbon-dioxide-free-and-the-calcium-carbonate-is-used-bythe-animal-to-form-its-shell-cauco-=-cacog-hoo-f-cothis-carbon-dioxide-was-originally-derived-from-the-air-5-dissociation-of-carbonates-by-heat-as-in-the-burning-oflime-this-is-a-matter-of-small-importance-especially-as-thelime-takes-up-the-carbon-dioxide-again-sooner-or-later-cacos-=-cao-co-carbon-dioxide-is-also-emitted-from-some-volcanoes-deepsprings-and-other-subterranean-sources-quantity-presentcountry-air-contains-on-an-av-2CJ846R.jpg)
Principles of agricultural chemistry [microform] . sea water is decomposed,setting carbon dioxide free, and the calcium carbonate is used bythe animal to form its shell. Ca(UCO,)., = CaCOg + HoO -f

Reductive calcination of calcium carbonate in hydrogen and methane: A thermodynamic analysis on different reaction routes and evaluation of carbon dioxide mitigation potential - ScienceDirect