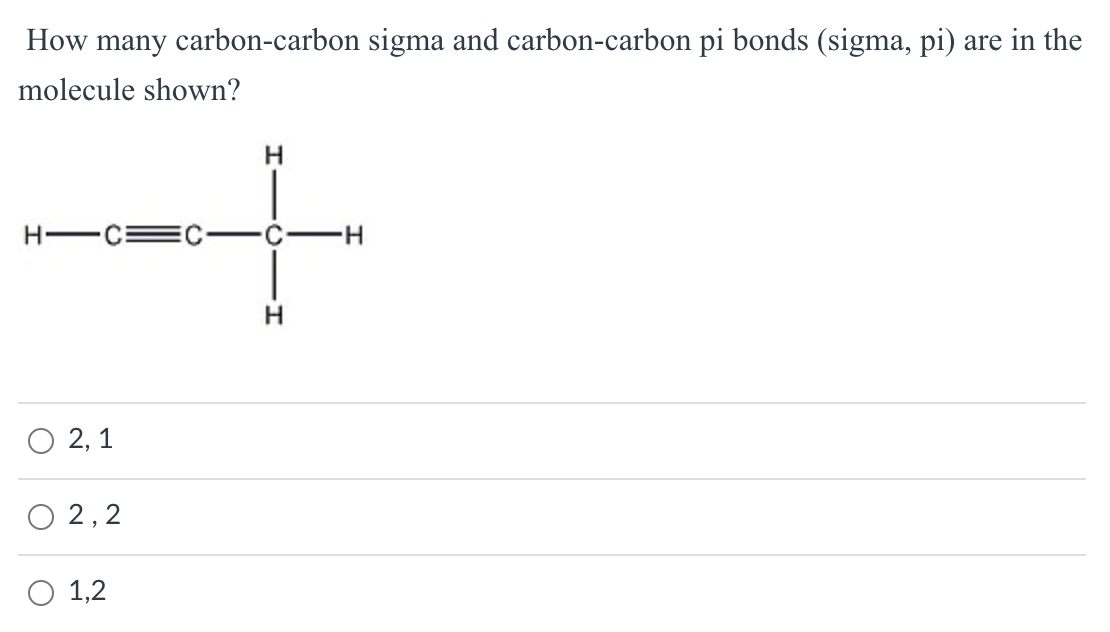
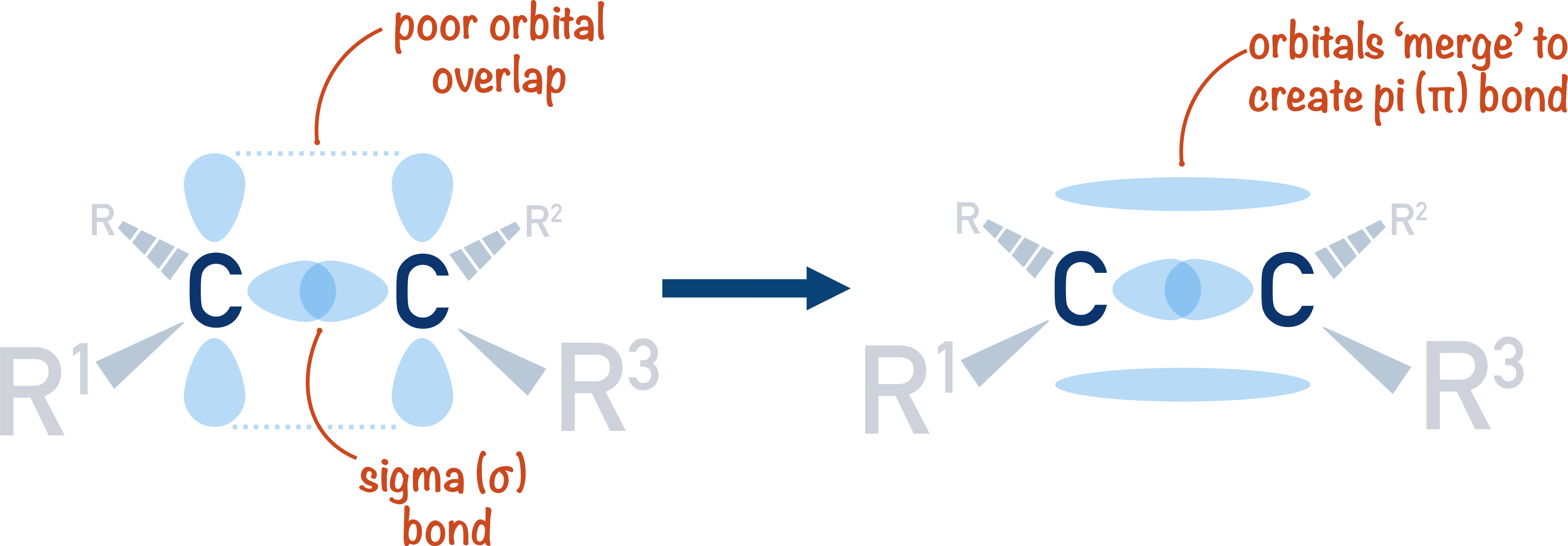
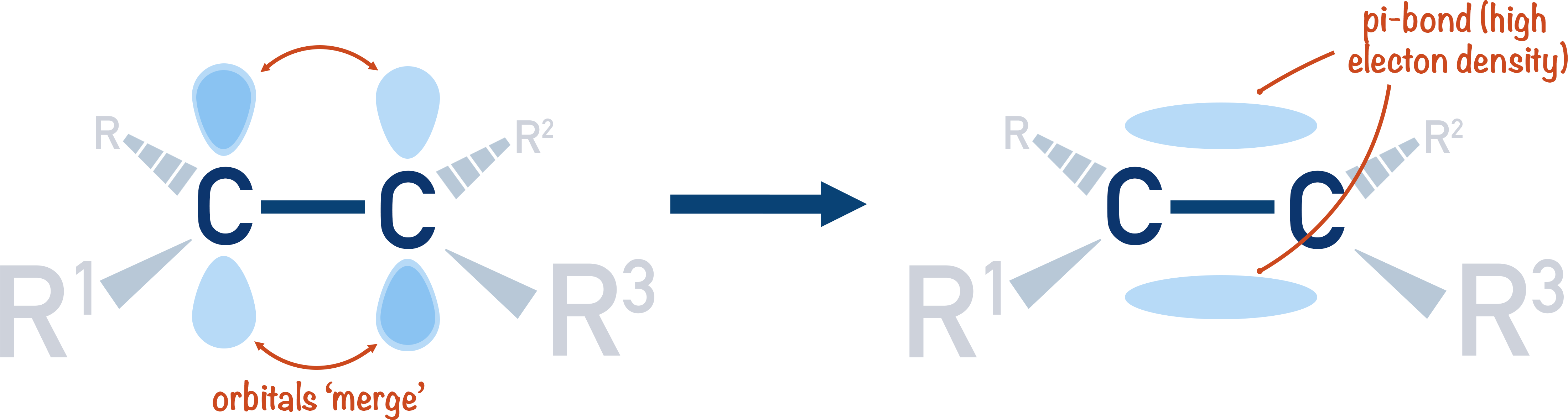
Draw a diagram to show only the hybrid orbitals around a single carbon atom in HBrCCBrH. Then draw a diagram to show all relevant orbitals (hybrid and otherwise) and label all bonds
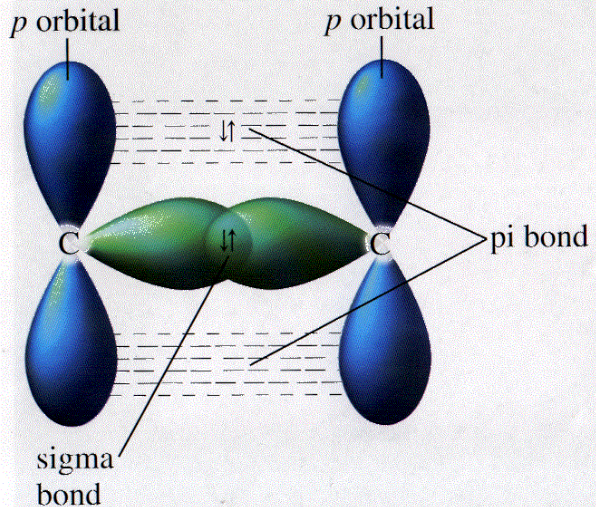
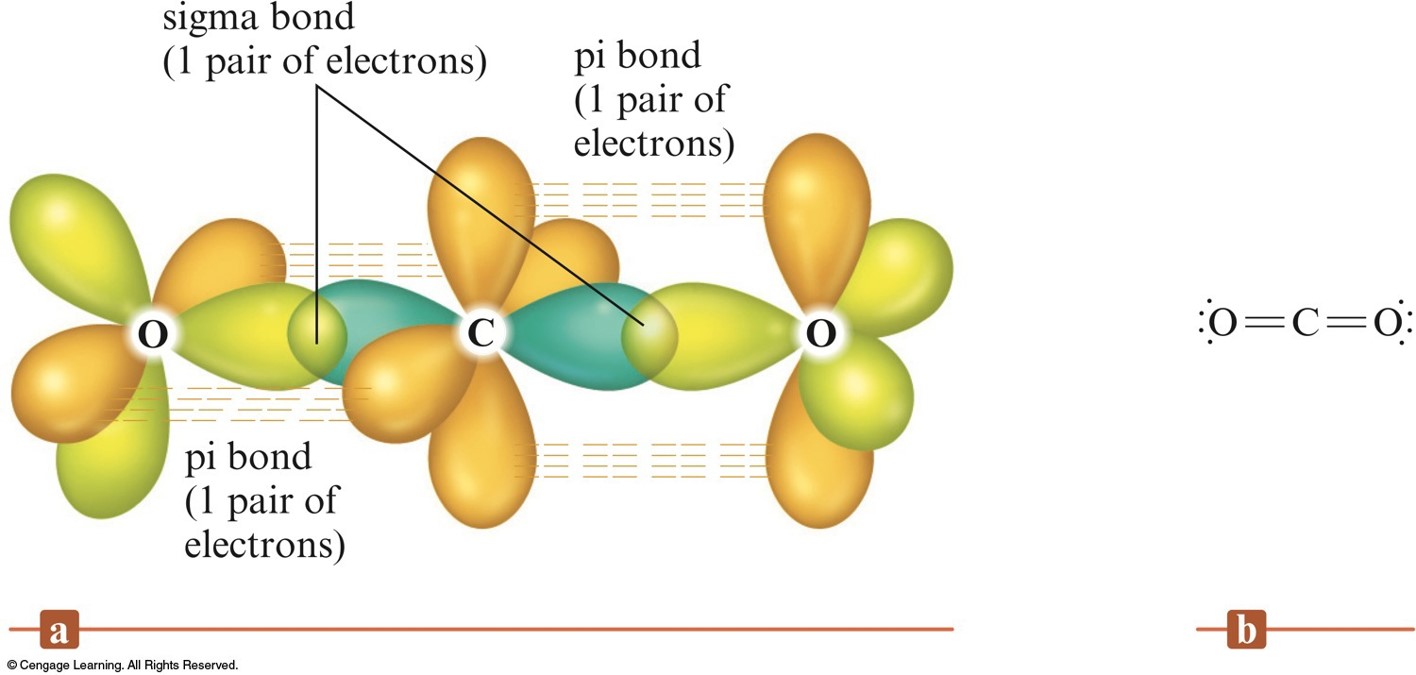
Which types of bonds are present between two carbon atoms in an acetylene molecule?Two sigma bonds and one pi bondThree pi bondsOne sigma bond and two pi bondsThree sigma bonds

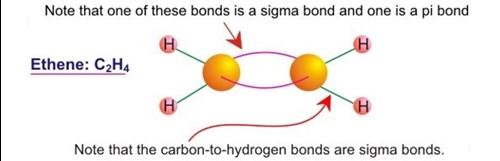
1.7.3.a. state the general formula of alkenes and understand that they are unsaturated hydrocarbons with a carbon-carbon double bond which consists of a sigma and a pi bond Flashcards | Quizlet
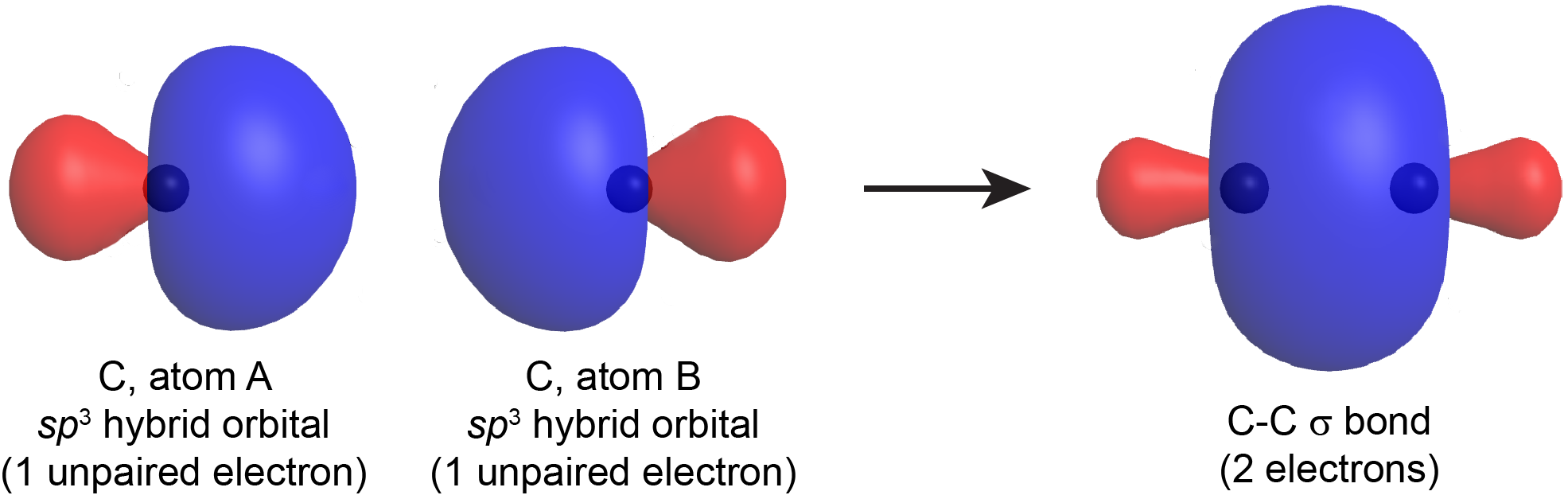
How does sigma/pi bond between two carbon atoms hold them together? When I pull a carbon atom how does the other carbon atoms move along with the carbon atom that is pulled? -
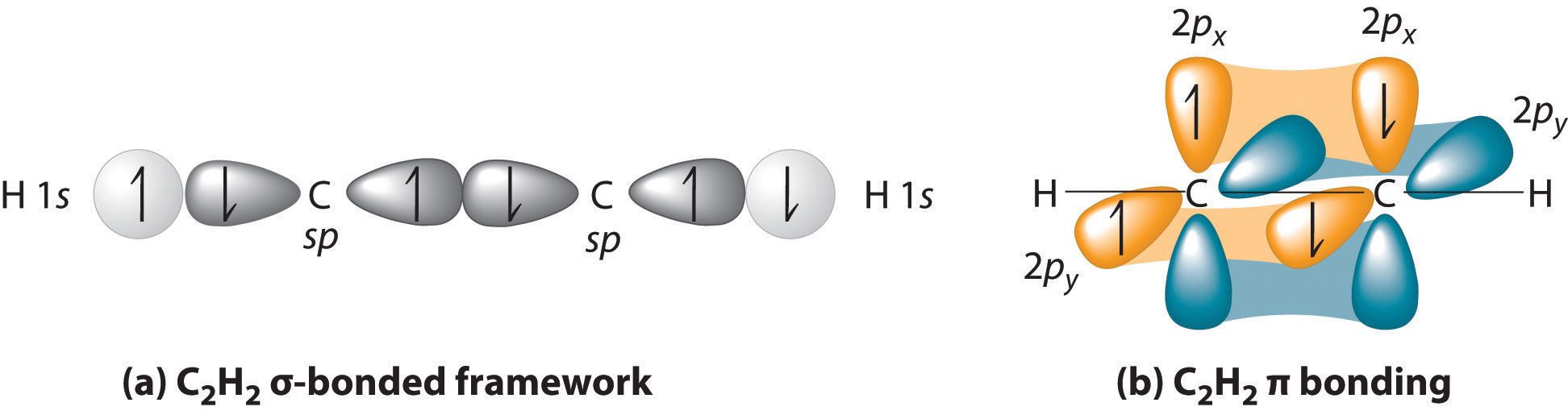
How does carbon use its "s" and "p" orbitals to form bonds in ethyne, ethene, and ethane? | Socratic



/chapter1/pages13and14/page13and14_files/chmo.png)