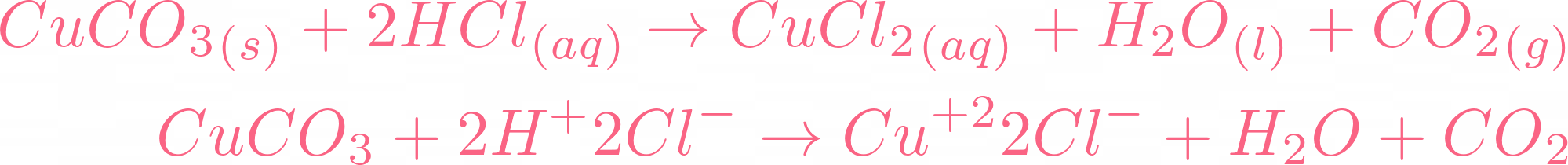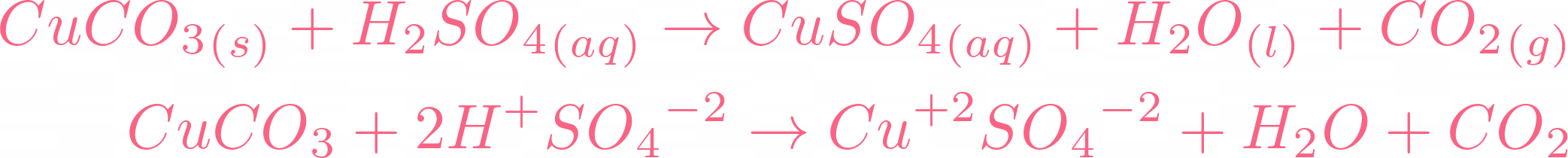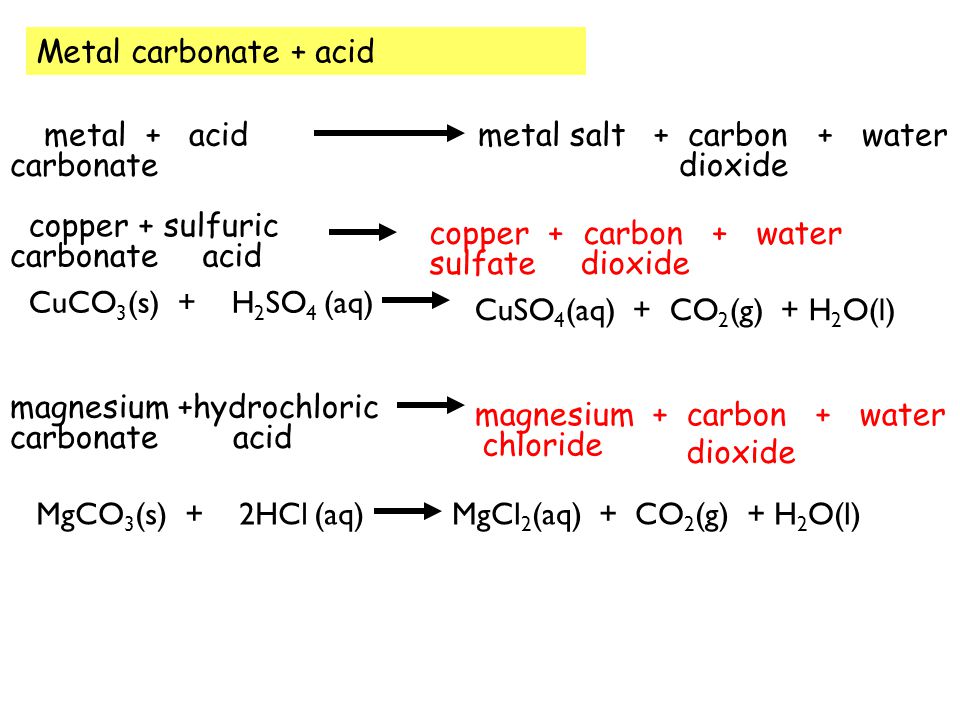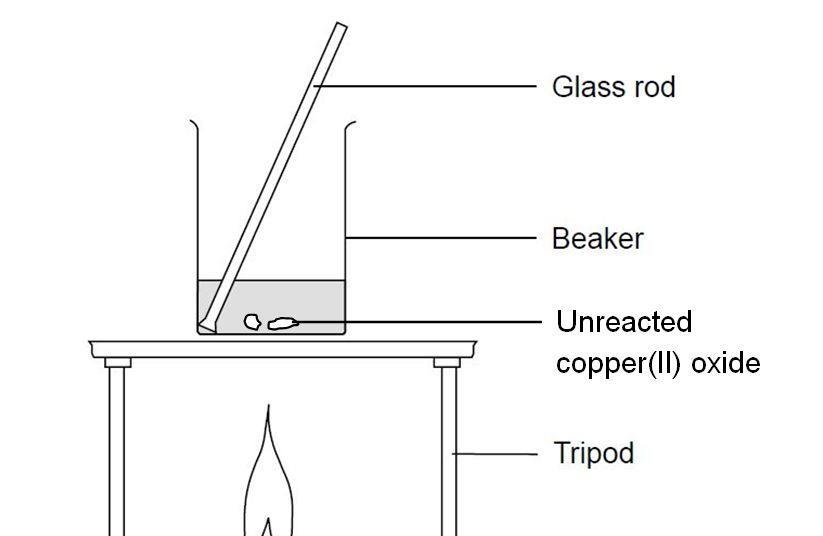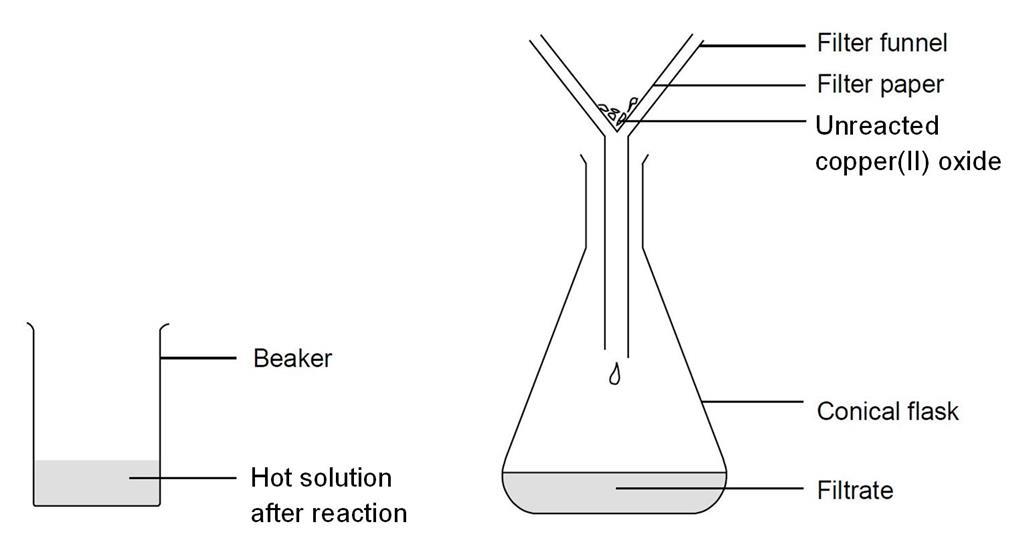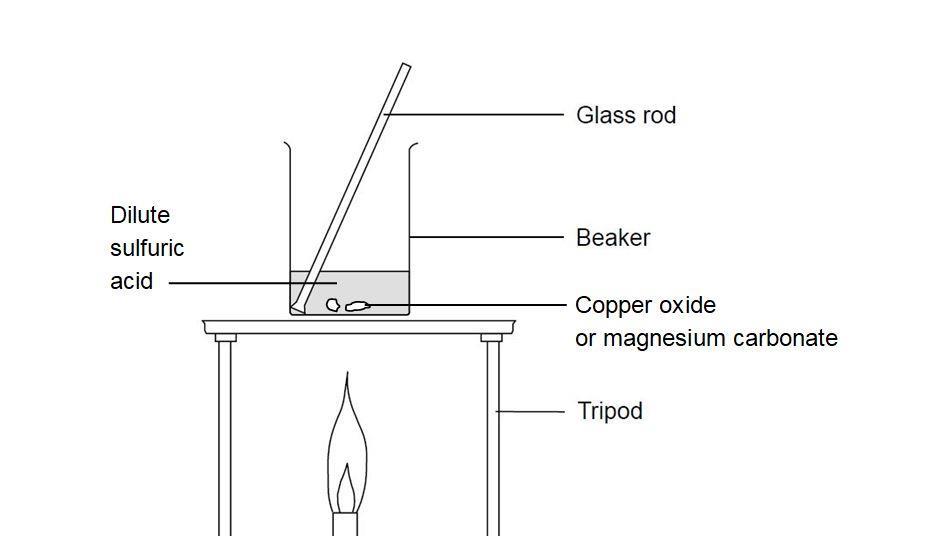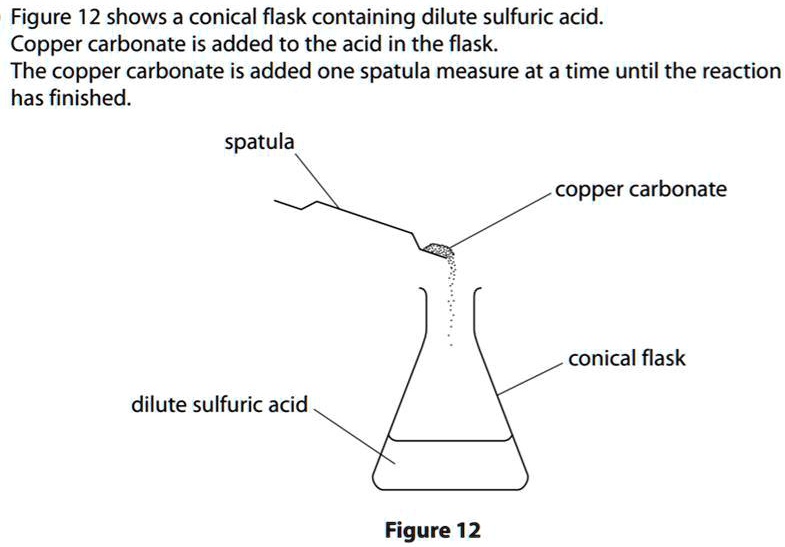
SOLVED: 'can someone please answer Figure 12 shows a conical flask containing dilute sulfuric acid. Copper carbonate is added to the acid in the flask The copper carbonate is added one spatula
![Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2021/05/mq2-3.jpg?w=640)
Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition

Most carbonates are insoluble (can not be dissolved in water) except those containing sodium or potassium ions. - ppt download