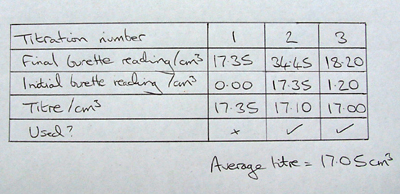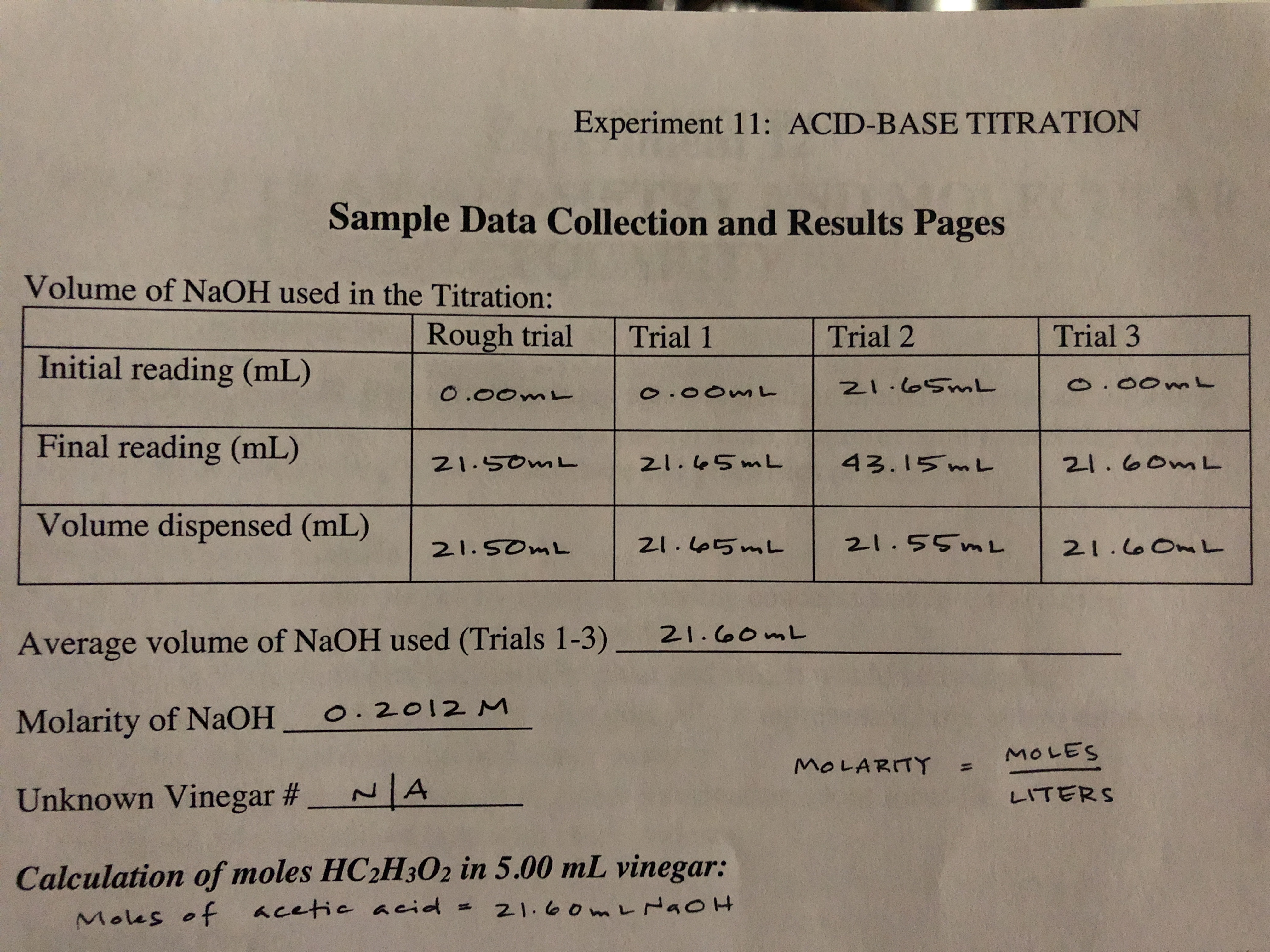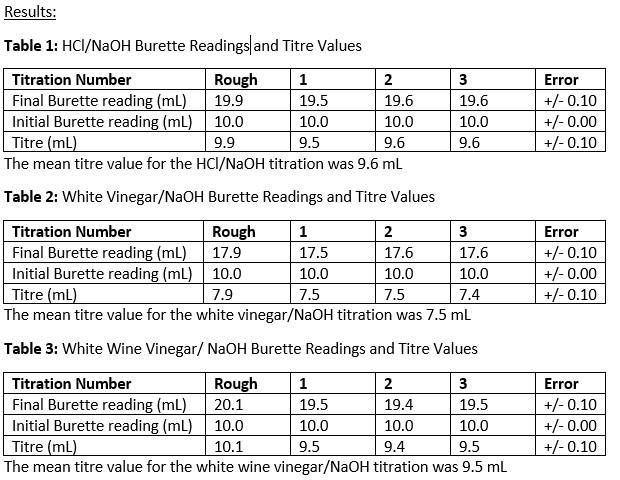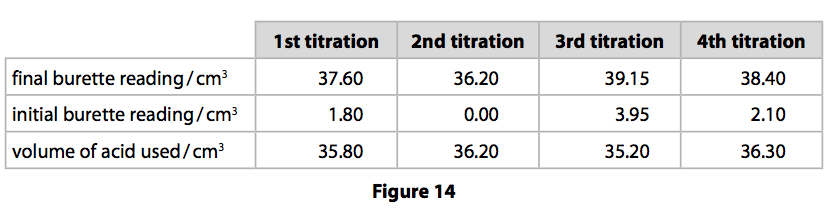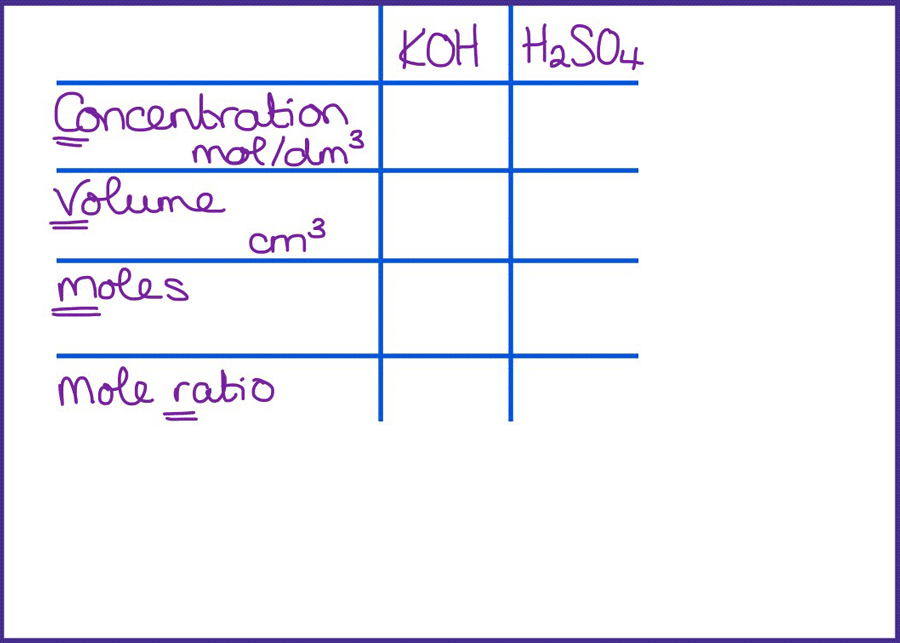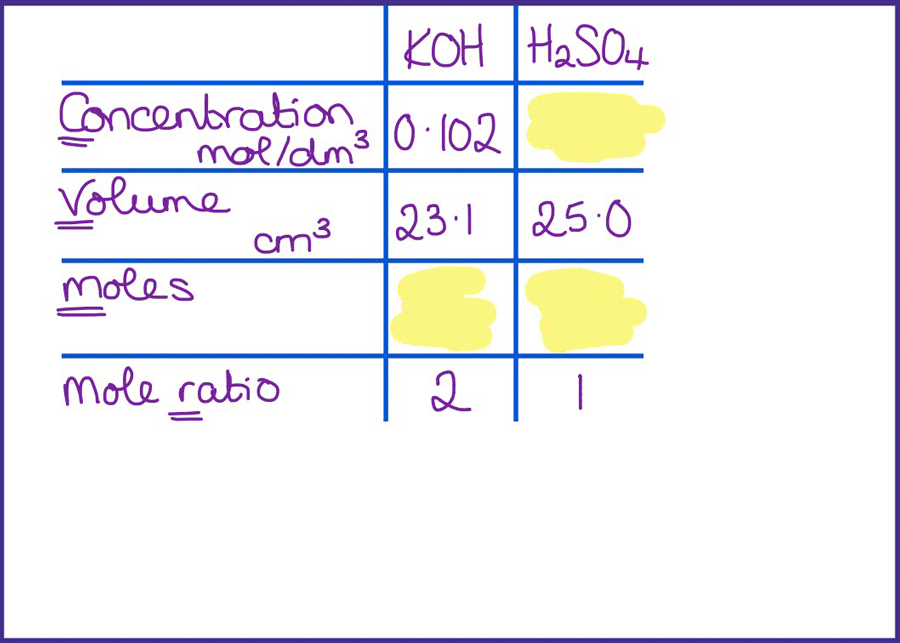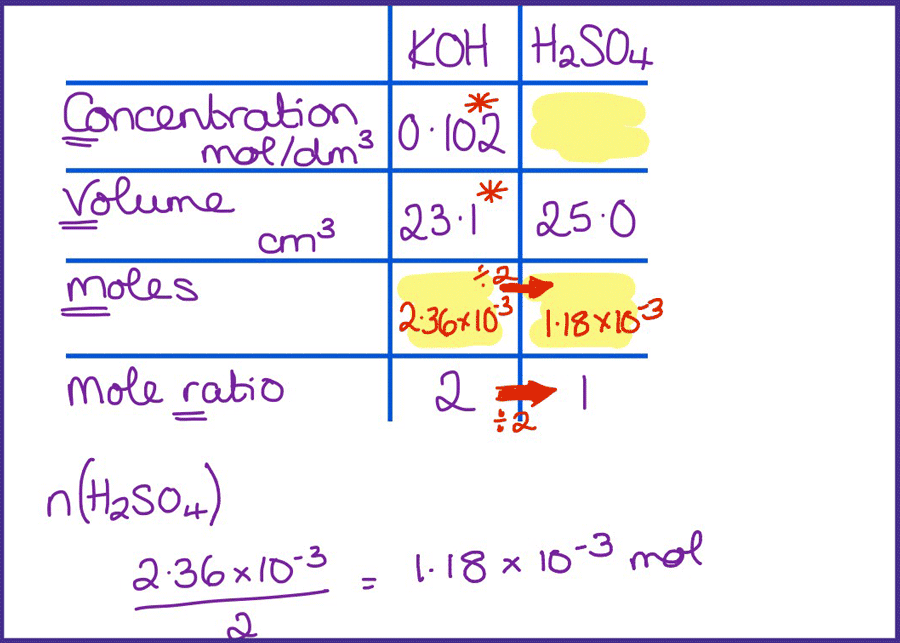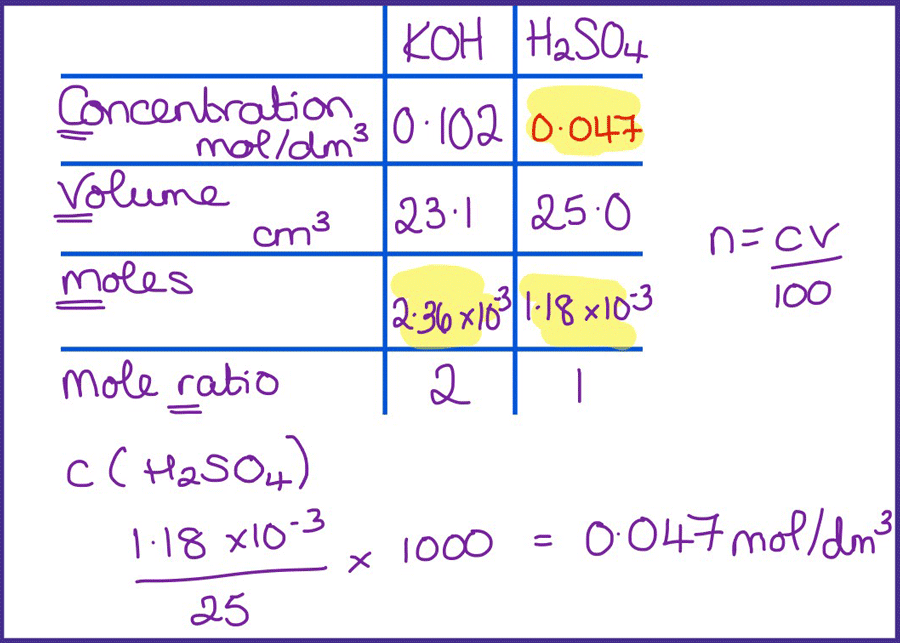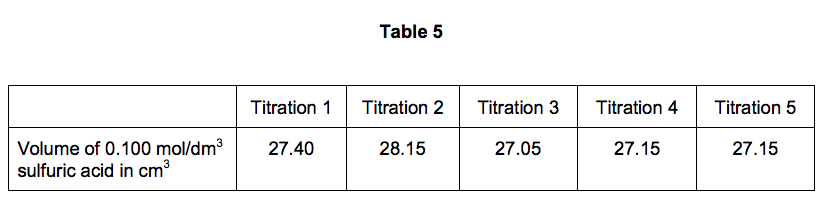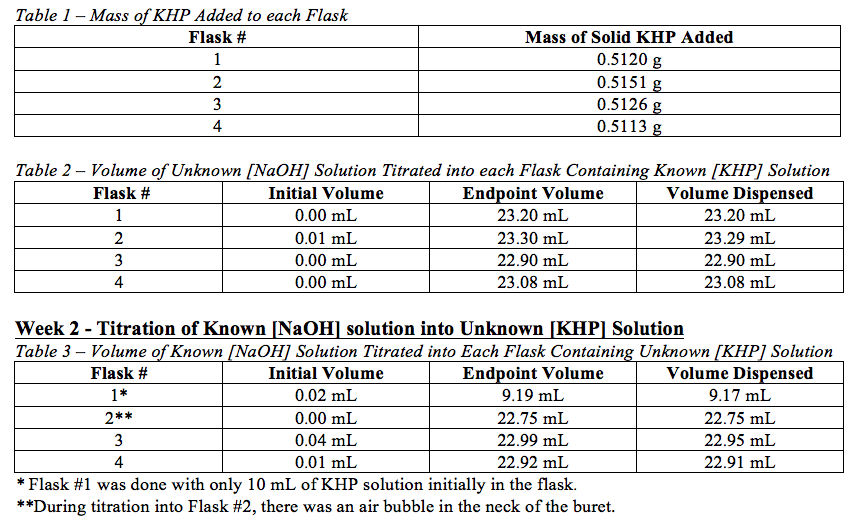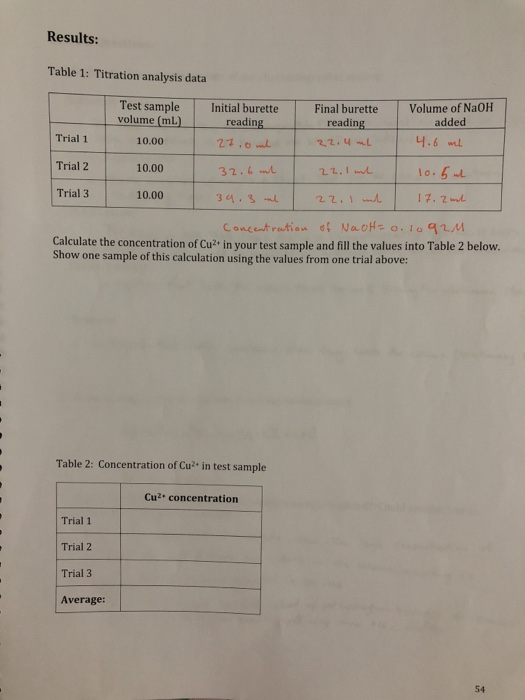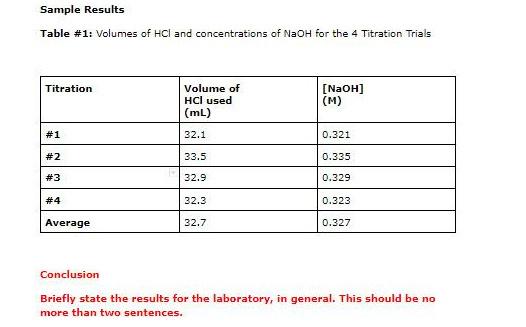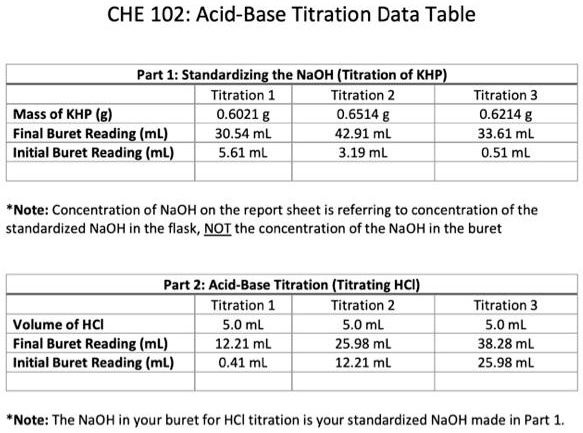
SOLVED: CHE 102: Acid-Base Titration Data Table Part 1: Standardizing the NaOH (Ttration of KHP) Titration Titration 2 Mass of KHP (g) 0.6021 g 0.6514 Final Buret Reading (mL) 30.54 mL 42.91

Table 2 from GREEN CHEMISTRY: STUDY OF ACID-BASE INDICATOR PROPERTY OF GOLDEN BEET ROOT | Semantic Scholar

Table 2 from GREEN CHEMISTRY: STUDY OF ACID-BASE INDICATOR PROPERTY OF GOLDEN BEET ROOT | Semantic Scholar
New analytical methodology for analysing S(IV) species at low pH solutions by one stage titration method (bichromatometry) with a clear colour change. Could potentially replace the state-of-art-method iodometry at low pH analysis